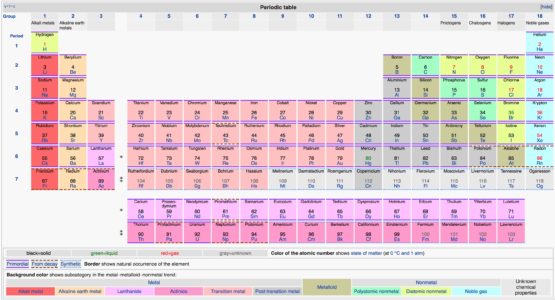
The nucleus of the atom gains protons moving from left to right, increasing the positive. For example, a quick glance at Figure AT5.2. Moving from left to right across a period, the atomic radius decreases. That way, we can get a better look at the relationship. Often it is useful to plot data on a graph. According to Coulombs Law, as the atomic number. - distance from the center of the nucleus to the outer edge of the electron cloud. As a result, fluorine is usually thought of as the most electronegative element. Such a relationship between atomic number and atomic radius is a direct correlation. However, on many scales, fluorine would be the most electronegative atom here. As a result, noble gases are also given electronegativity values on this scale. The Allen scale just depends on the ability of an atom to interact with light, which is something even noble gases can do. Major periodic trends include atomic radius, ionization energy, electron affinity, electronegativity, valency and metallic character. They were discovered by the Russian chemist Dmitri Mendeleev in the year 1863. There are some small exceptions, such as the oxygen radius being slightly greater than the nitrogen radius. Periodic trends are specific patterns that are present in the periodic table that illustrate different aspects of a certain element. Periodic Trend The atomic radius of atoms generally decreases from left to right across a period. Some electronegativity scales do not have values for the noble gases, because they are based on experimental measurements of compounds, and noble gases do not commonly form compounds with other elements. Atomic radii of the representative elements measured in picometers. 
\): The Allen electronegativity values of the second-row elements. So mass should increase across periodic table rows from left to right and down periodic table columns experimental evidence supports this prediction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
